Blog
Immunoassay Screening: Principles, Accuracy & Drug Testing

TL;DR:
- Immunoassay screening is a sensitive, rapid, and scalable method for initial drug detection but has limitations.
- Confirmatory testing with mass spectrometry is essential due to potential false positives and blind spots.
- Proper interpretation, panel selection, and ongoing review are critical for reliable drug testing practices.
Immunoassay screening is the preliminary method of choice for most workplace and forensic drug tests, yet practitioners regularly encounter results that don’t tell the full story. A reactive screen can reflect a legitimate exposure, a cross-reacting medication, or a synthetic compound the assay was never designed to catch. Speed and scalability are real advantages, but they come with performance boundaries that every forensic scientist, clinician, and law enforcement professional must understand before acting on a result. This guide breaks down the biochemical mechanics, major assay formats, real-world accuracy benchmarks, and expert protocols for making immunoassay data work reliably in professional settings.
Table of Contents
- Understanding immunoassay screening: How it works and test mechanics
- Major immunoassay formats: From EMIT to lateral flow
- Accuracy, cutoffs, and key limitations in practice
- Expert strategies for interpreting and confirming results
- The practical truth: Why immunoassay screening is irreplaceable—yet never enough
- Trusted solutions for accurate and efficient drug screening
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Rapid screening tool | Immunoassay screening offers fast, high-throughput initial drug testing across many settings. |
| Limited accuracy | False positives and negatives require confirmation by mass spectrometry for legal or clinical reliability. |
| Panel-dependent results | Detection scope is limited by the drugs or metabolites targeted on each immunoassay panel. |
| Cutoffs and cross-reactivity | Interpreting immunoassay screens demands understanding official cutoffs and the risks of cross-reactivity with medications. |
| Expert judgment needed | Effective drug testing combines rapid screening with thoughtful result interpretation and appropriate follow-up testing. |
Understanding immunoassay screening: How it works and test mechanics
Immunology and chemistry meet at the core of every immunoassay. The test uses antibodies engineered to recognize specific drug molecules or their metabolites present in a biological sample, most commonly urine, though saliva and blood are also used. When a sample is introduced, a competition happens between the drug in the sample and a labeled version of that same drug for available antibody binding sites.
The signal produced—whether optical color intensity or enzyme activity—is inversely related to drug concentration. More drug in the sample means less labeled compound binds, which means a weaker signal. Once concentration crosses a pre-set cutoff, the test reports a presumptive positive. It is qualitative, not quantitative. You get above or below the threshold, not a precise concentration.
Key mechanics at a glance:
- Sample types: Urine (most common), oral fluid, whole blood
- Detection target: Drug molecules or primary metabolites
- Result logic: Signal inversion relative to analyte concentration, as described in competitive binding principles
- Output: Presumptive positive or negative based on cutoff
- Regulatory standard: SAMHSA sets reporting cutoffs, for example THC at 50 ng/mL for urine
Understanding toxicology screening basics helps contextualize why cutoffs exist: they balance sensitivity (catching true positives) against specificity (avoiding false alarms). A cutoff set too low catches more true users but also more innocent cross-reactors. Set too high, and low-level exposures slip through.
Statistic callout: SAMHSA-mandated urine cutoffs include amphetamines at 500 ng/mL, cocaine metabolite at 150 ng/mL, and THC at 50 ng/mL. These thresholds define the floor for federal workplace programs.
Pro Tip: When evaluating a new panel for your program, always request the manufacturer’s antibody cross-reactivity data alongside the cutoff table. That document reveals far more about real-world performance than sensitivity claims alone.
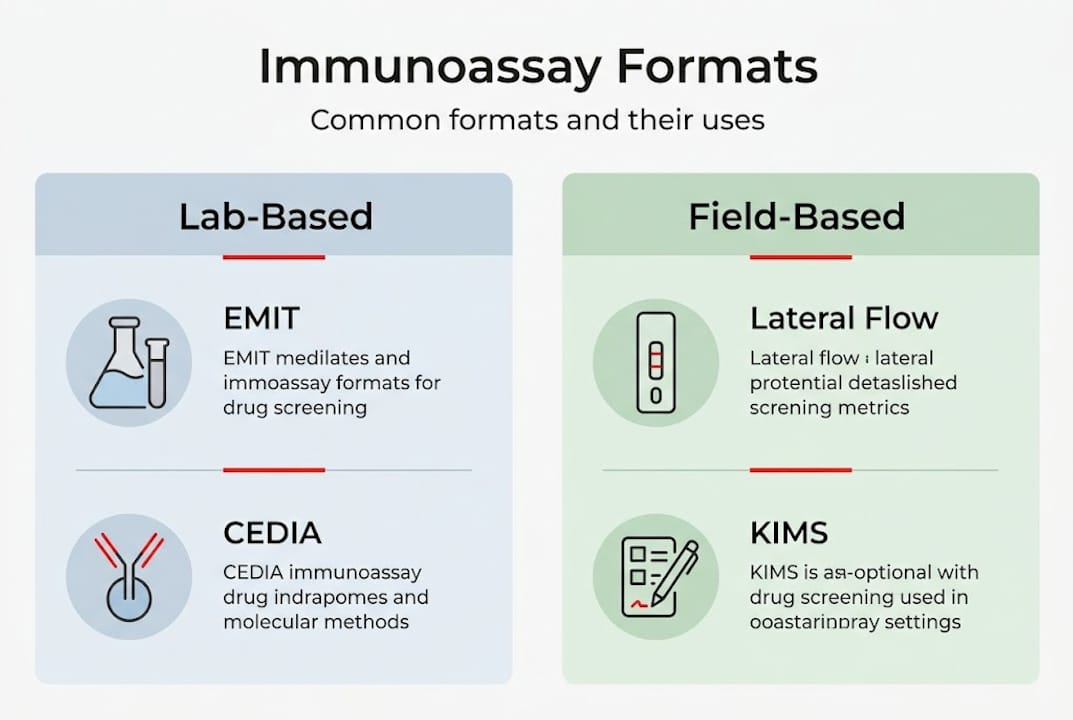
Major immunoassay formats: From EMIT to lateral flow
Not all immunoassays are built the same. The format you choose shapes throughput, sensitivity, infrastructure requirements, and cost per test. Here is how the leading technologies compare.
| Format | Mechanism | Best use case | Throughput |
|---|---|---|---|
| EMIT | Enzyme activity modulated by drug binding | Central lab, high volume | High |
| CEDIA | Enzyme fragment recombination | Automated lab platforms | Very high |
| KIMS | Microparticle aggregation inhibition | Large-scale urine screens | High |
| ELISA/CLIA | Microplate antibody binding, optical read | High-sensitivity confirmation or research | Medium |
| LFA (lateral flow) | Membrane-based competitive flow | Point-of-care, field, onsite | Low to medium |
Leading techniques including EMIT, CEDIA, KIMS, and LFAs each occupy a specific niche. EMIT and CEDIA dominate high-throughput clinical and forensic labs because they integrate with automated analyzers and process hundreds of samples per hour. KIMS uses microparticle aggregation to generate a signal change, making it robust for large-scale urine screening programs.
Lateral flow assays (LFAs) are the technology behind most cup and cassette formats used in rapid screening methods. They require no instrumentation, deliver results in minutes, and are CLIA waived for point-of-care use. That makes them ideal for probation offices, emergency departments, and onsite employer programs.
Different technologies support high-throughput and rapid POC screening, but selection should always match the setting:
- High-volume lab: EMIT or CEDIA on automated platforms
- Field or onsite: Lateral flow cups or cassettes
- Research or confirmatory support: ELISA or CLIA formats
- Emerging drug surveillance: Panels with updated antibody targets
Pro Tip: If your program operates across both field and lab settings, standardize cutoffs across formats. Discrepancies between a field LFA and a lab EMIT result on the same sample are often a cutoff mismatch, not a test failure.
Accuracy, cutoffs, and key limitations in practice
Immunoassay performs well for the drug classes it was designed to detect, but real-world accuracy is more complicated than sensitivity percentages suggest. Every assay has a performance ceiling shaped by antibody specificity, cutoff calibration, and the chemistry of the sample matrix.
Key SAMHSA urine cutoffs (federal programs):
| Drug class | Initial test cutoff | Confirmatory cutoff |
|---|---|---|
| THC metabolite | 50 ng/mL | 15 ng/mL |
| Amphetamines | 500 ng/mL | 250 ng/mL |
| Cocaine metabolite | 150 ng/mL | 100 ng/mL |
| Opiates | 2,000 ng/mL | 2,000 ng/mL |
| PCP | 25 ng/mL | 25 ng/mL |
These SAMHSA/HHS cutoffs for urine represent the federal standard, but clinical and forensic programs may apply different thresholds based on context.
Where immunoassay falls short:
- Cross-reactivity causing false positives. Antibodies recognize structural similarities, not exact molecules. Rifampin can trigger opiate positives. Certain antihistamines may flag amphetamines. False positives can reach 34% for opiates in some study populations.
- Synthetic drug blind spots. Fentanyl, many designer benzodiazepines, and novel psychoactives often evade standard panels because the antibodies were not developed for those structures.
- False negatives at low concentrations. Drugs near but below the cutoff, or those that metabolize slowly, may not trigger a positive even when use occurred.
- Matrix interference. Adulterants, dilution, or abnormal pH can suppress signal and produce false negatives.
“All immunoassay positives are presumptive. No forensic or clinical decision should rest on an unconfirmed screen.”
Understanding false positives in drug tests is not optional for professionals working in legal or evidentiary contexts. Similarly, drug testing accuracy depends on both assay design and proper result interpretation. Always review how to interpret drug test results before acting on any screen.
Expert strategies for interpreting and confirming results
Knowing what immunoassay can and cannot do is only half the equation. The other half is building a workflow that accounts for its limitations without creating operational paralysis.
Core protocols every program should follow:
- Treat every positive as presumptive. Presumptive positives must be confirmed with GC/MS or LC-MS/MS before any adverse action, legal proceeding, or clinical intervention.
- Report language matters. Document results as “presumptive positive, unconfirmed” until mass spectrometry verification is complete. This protects both the subject and the program.
- Expand panels for current drug trends. Polysubstance use, emerging synthetics, and pediatric or trauma exceptions demand flexible panel selection. A standard 5-panel misses fentanyl, synthetic cannabinoids, and many designer drugs entirely.
- Contextualize interpretation. A workplace screen operates under different standards than a pediatric toxicology case or a forensic death investigation. Adjust cutoffs and interpretation accordingly.
- Document everything. For evidentiary use, record which assay was used, the cutoff applied, the result, and the rationale for any follow-up action.
Factors that influence accuracy go beyond the test strip itself. Review factors influencing accuracy when auditing your program, and revisit selecting test panels whenever your population’s drug use patterns shift.
Pro Tip: Build a standing review cycle into your program, at minimum annually, to assess whether your current panel still reflects the substances actually present in your population. Emergency department toxicologists and forensic labs in your region are excellent sources for local drug trend data.
The practical truth: Why immunoassay screening is irreplaceable—yet never enough
After years of working with drug testing programs across forensic, clinical, and workplace settings, one thing stands out: immunoassay is the best triage tool available, and it is routinely asked to do more than triage.
The speed and scalability of lateral flow and automated immunoassay platforms are genuinely unmatched. No other technology lets you screen hundreds of samples per shift at a cost that makes population-level surveillance feasible. That value is real and should not be dismissed.
But the polysubstance era has changed the math. High co-use rates, with cocaine at 34% and fentanyl at 17% in emergency department populations, mean that a clean screen no longer provides the reassurance it once did. A patient or subject may be using multiple substances, some of which the panel simply cannot see.
The mindset shift needed is this: treat immunoassay as the beginning of an investigation, not its conclusion. Expand panels proactively. Invest in confirmation infrastructure or partnerships with reference labs. And resist the organizational pressure to act on a screen alone when the stakes are high.
Learn more about forensic drug testing importance to understand why confirmation is not a bureaucratic formality but a scientific and legal necessity.
Trusted solutions for accurate and efficient drug screening
Professionals who understand immunoassay’s strengths and limits need testing tools that are built to match. At rapidtestcup.com, we stock CLIA waived, FDA-approved kits designed for high-volume forensic, clinical, and workplace programs.
Our urine drug test kits comparison helps you identify the right format for your throughput and panel requirements. If you are building or updating a program from scratch, the guide to selecting drug test kits walks through every decision point. For programs that need expanded coverage, the 18-panel economy drug test cup covers major and emerging substances including fentanyl, K2, and ETG, with built-in adulterant checks.
Frequently asked questions
What does immunoassay screening test for in drug testing?
Immunoassay screens detect drug classes or metabolites including amphetamines, cocaine, opiates, cannabis, PCP, and more, depending on the specific panel selected.
How accurate is immunoassay screening for drugs?
Immunoassay is highly sensitive but can produce false positives up to 34% in some drug classes and may miss synthetics or very low concentrations without mass spectrometry confirmation.
Why do immunoassay results need confirmation by GC/MS or LC-MS/MS?
Because immunoassay is a presumptive test subject to cross-reactivity, confirmatory mass spectrometry is required for definitive identification in clinical or legal contexts.
What causes false positives on immunoassay drug screens?
Substance cross-reactivity from prescription or OTC medications, such as certain antibiotics or cold remedies, can trigger false positives for specific drug classes.
Which drugs are most likely to be missed by immunoassay screening?
Synthetic drugs missed by immunoassay include fentanyl, designer benzodiazepines, and novel psychoactives not specifically targeted by the panel’s antibody design.