Blog
Adulterant Detection Methods for Accurate Drug Testing

TL;DR:
- Adulteration methods include dilution, chemical agents, substitution, and internal adulterants.
- Specimen validity testing detects tampering through parameters like creatinine, pH, specific gravity, and oxidants.
- Advanced tools such as mass spectrometry and proteomics help identify sophisticated or degraded adulterants.
Adulterant Detection Methods for Accurate Drug Testing
Specimen tampering is more common than most programs want to admit. Studies confirm that common adulteration methods like dilution, chemical agents, and synthetic urine are increasingly sophisticated, and many collection programs are still relying on detection strategies designed for a simpler era. A single missed adulteration event can corrupt a chain of custody, void a legal proceeding, or allow a dangerous individual to pass a workplace screen. This article breaks down the full spectrum of adulterant detection methods, from basic strips to mass spectrometry and proteomics, so you can build a protocol that holds up under pressure and regulatory scrutiny.
Table of Contents
- What is adulteration in drug testing?
- How specimen validity testing (SVT) works: The compliance backbone
- Tiered detection methods: From strips to mass spectrometry
- Limits, loopholes, and the emerging science
- Expert perspective: What most protocols still miss about adulterant detection
- Reliable adulterant detection starts with advanced tools
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Adulteration is sophisticated | Today’s adulterants can outpace routine detection, requiring layered strategies. |
| SVT is foundational | Specimen validity testing is a regulatory requirement that flags tampering early. |
| Tiered methods work best | Combining rapid strips, immunoassay, and lab confirmation balances cost, speed, and accuracy. |
| Limits exist | No single test catches all adulterants, especially emerging synthetics and detox agents. |
| Continuous vigilance needed | Regular protocol updates and advanced technology adoption are crucial to maintain integrity. |
What is adulteration in drug testing?
Adulteration refers to any deliberate action taken to falsify, mask, or alter the outcome of a urine drug test. The goal is always the same: make a positive sample appear negative, or render the specimen untestable. What has changed dramatically over the past decade is the sophistication of the methods being used.
Common adulteration methods fall into four major categories: dilution, chemical addition, substitution, and internal adulterants. Each creates a distinct set of challenges for detection.
Dilution is the most accessible method. The donor drinks excessive fluids before the test, lowering drug metabolite concentrations below detectable thresholds. In-collection dilution involves adding water directly to the specimen cup. Both tactics are common, and both are detectable with proper specimen validity testing.
Chemical addition targets the immunoassay chemistry itself. Agents like household bleach, vinegar, nitrites (sold commercially as “Urine Luck”), and oxidizing compounds such as pyridinium chlorochromate (sold as “Klear”) are introduced directly into the specimen. These chemicals either destroy the drug molecules or interfere with the antibody-antigen reaction the immunoassay depends on.
Substitution means replacing urine entirely. This includes synthetic urine products, which have become chemically sophisticated enough to mimic real urine’s creatinine, pH, and specific gravity. It also includes using someone else’s urine, often stored in a hidden container or bladder device.
Internal adulterants are ingested before testing. Certain detox drinks or high-dose niacin supplements claim to flush or mask drug metabolites, though their actual effectiveness varies by substance, timing, and metabolism.
Understanding the tools and methods for detection that counter each category is essential for any serious program. Here is a quick breakdown of the primary adulteration tactics:
- Drinking excess water before collection (internal dilution)
- Adding tap water or other fluids directly to the specimen
- Introducing chemical agents like bleach, nitrites, or oxidants
- Submitting synthetic urine or a third party’s clean urine
- Ingesting commercial detox products that affect metabolite clearance
The legal stakes are not trivial. Per regulatory guidance, an adulterated sample equals refusal, which carries the same consequences as a confirmed positive result in most regulated programs. In DOT-regulated industries, this means immediate removal from safety-sensitive duties.
“An adulterated or substituted specimen result is reported as a refusal to test, which is treated identically to a verified positive result under federal workplace drug testing policy.”
Consider this scenario: a transportation employee dilutes their specimen sufficiently to drop amphetamine metabolites below the 500 ng/mL cutoff. Without creatinine and specific gravity screening, that specimen passes. The driver returns to work. The liability exposure is significant, and the impact of adulterants on program integrity is direct and measurable.
How specimen validity testing (SVT) works: The compliance backbone
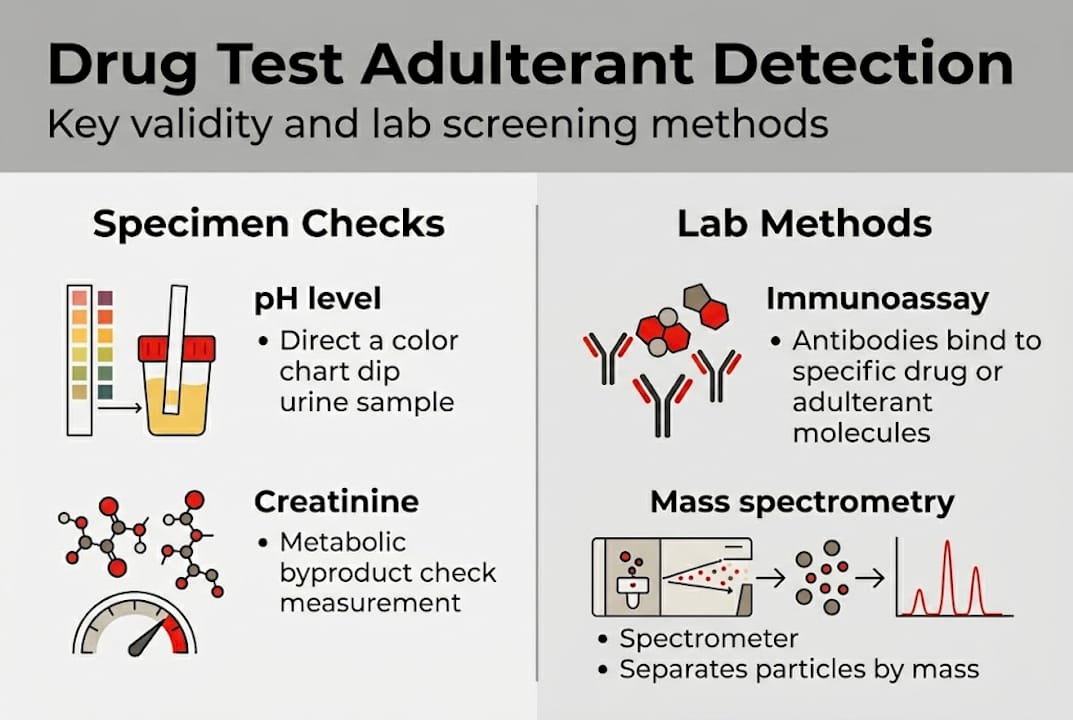
Specimen validity testing is the regulatory and scientific foundation on which all trustworthy drug testing programs are built. Before any immunoassay or confirmatory test runs, SVT screens the specimen itself to verify that what you received is actually testable, human urine.
SAMHSA/HHS guidelines mandate that certified laboratories conduct SVT as a prerequisite to drug analysis. The parameters are specific and non-negotiable for federally mandated testing: color, odor, foam, pH, creatinine, specific gravity, and screens for oxidants, surfactants, and other adulterant-specific markers.
Here is what each parameter reveals:
pH detects acid or base addition. Normal urine ranges from 4.5 to 8.5. A pH below 3 or above 11 indicates chemical tampering. Vinegar drives pH sharply acidic; bleach or ammonia drives it alkaline.
Creatinine is a metabolic waste product produced at a relatively constant rate by muscle metabolism. Normal creatinine is 20 to 300 mg/dL. Values below 2 mg/dL, combined with specific gravity below 1.001, flag the specimen as substituted. Values between 2 and 20 mg/dL signal dilution.
Specific gravity measures the density of solutes in urine. Normal range is 1.003 to 1.030. A reading of exactly 1.000 or 1.001 on a diluted specimen with low creatinine points directly to substitution or extreme dilution.
Oxidant screens detect the presence of chemicals like nitrites, chromate, or bleach. Nitrite levels above 500 mcg/mL are considered adulterated per federal guidelines.
| Parameter | Normal Range | Abnormal Interpretation |
|---|---|---|
| pH | 4.5 to 8.5 | Below 3 or above 11: chemical addition |
| Creatinine | 20 to 300 mg/dL | Below 2: substituted; 2 to 20: diluted |
| Specific gravity | 1.003 to 1.030 | Below 1.001: substituted or extreme dilution |
| Nitrites | Below 200 mcg/mL | Above 500 mcg/mL: adulterated |
| Oxidants/chromate | Negative | Any presence: adulterated |
For ensuring specimen validity under federal and workplace standards, SVT must be treated as the first gate, not an afterthought.
Pro Tip: Run SVT before committing to a full immunoassay panel. A specimen flagged as substituted or adulterated at this stage should be reported immediately rather than proceeding to drug-specific testing, which can produce misleading data on an invalid matrix.
SVT also plays a practical role in court defensibility. When a specimen is reported as adulterated, that report is based on validated chemical parameters with defined cutoffs, not subjective observation. That precision matters when a positive equivalent result is challenged legally.
Tiered detection methods: From strips to mass spectrometry
Validating the specimen is only the first gate. The actual detection of adulteration agents and drug metabolites requires a layered approach where each method serves a specific role in the testing sequence.
The tiered framework works like this: rapid strips at the point of collection, laboratory immunoassay for screening, gas chromatography/mass spectrometry (GC/MS) or liquid chromatography/mass spectrometry (LC/MS/MS) for confirmation, and proteomics for contested or sophisticated cases.
Adulterant strips detect common agents quickly and cheaply but have limited scope. Mass spectrometry confirms drug presence with legal defensibility, while proteomics advances detection into novel territory by analyzing protein and peptide changes caused by chemical manipulation.
Here is a comparison of the four primary tiers:
| Method | Cost | Speed | Coverage | Limitations | Best Use |
|---|---|---|---|---|---|
| Rapid strip | Low | Immediate | Common adulterants | Misses novel agents | Point of collection screening |
| Immunoassay | Moderate | Hours | Broad drug classes | Cross-reactivity, false negatives | Lab screening |
| GC/MS or LC/MS/MS | High | 24 to 72 hours | Specific compounds | Requires lab infrastructure | Confirmatory testing |
| Proteomics | Very high | Days | Manipulation signatures | Research stage, limited commercial availability | Highly contested specimens |
For test strip effectiveness in field environments, multi-adulterant strips cover pH, nitrites, glutaraldehyde, bleach, creatinine, and specific gravity simultaneously. These are practical and fast, but they cannot identify every novel chemical agent entering the market.
When adulteration is suspected at the collection stage, follow a structured escalation path:
- Document all observable signs: temperature out of range (normal is 90 to 100°F within 4 minutes of collection), unusual color, odor, or foam.
- Run rapid adulterant strips immediately if your protocol includes them at collection.
- Flag the specimen for SVT before proceeding to immunoassay.
- If SVT parameters are borderline or inconsistent, escalate to confirmatory GC/MS or LC/MS/MS testing.
- For specimens under legal challenge where tampering is suspected but unconfirmed, request proteomics analysis if available through your reference laboratory.
For interpreting results across these tiers correctly, understanding which method applies to which question is critical. Strips answer “is something wrong here?” Mass spectrometry answers “what exactly is present and at what concentration?” Proteomics answers “was this specimen chemically manipulated, even if the adulterant itself is no longer detectable?”
Pro Tip: Rapid strips are screening tools, not final arbiters. If a strip result conflicts with observable specimen characteristics, the specimen should proceed to full lab evaluation rather than being cleared based on the strip result alone.
Limits, loopholes, and the emerging science
No detection system catches everything, and sophisticated donors know it. Understanding the current gaps in detection is as important as understanding the tools themselves.
The most significant limitation of field-based methods is their fixed scope. Rapid strips are calibrated against known adulterants. When a new oxidizing agent or enzyme inhibitor enters the market, it may not trigger existing test chemistry. Laboratories playing catch-up is a recurring pattern in forensic toxicology.
Novel adulterants and some detox agents may evade common screens, but proteomics can uncover hidden manipulations by detecting altered protein and peptide signatures in the specimen matrix. This is significant because it shifts the detection target from the adulterant itself to the biological consequence of its presence.
Current loopholes and limitations worth tracking include:
- Oxidizing agents that degrade before lab analysis, leaving no chemical trace
- Detox products formulated to temporarily normalize creatinine and specific gravity values while reducing drug metabolite concentrations
- Synthetic urine products updated to include uric acid, biocides, and buffering agents that mimic real urine chemistry closely
- Certain synthetic urine risks are especially relevant for programs without direct observation protocols
- Fermentation artifacts from delayed specimen processing that alter pH and creatinine readings, creating false adulteration flags
On the immunoassay side, false negatives for certain drugs can be as high as 40% when interference agents are present in the specimen, which is a significant vulnerability for programs relying solely on screening without confirmatory follow-up.
Staying informed about emerging trends in forensic toxicology and adulteration science is not optional for serious programs. The technology being used to beat drug tests evolves continuously, and so must the methods used to counter it.
“Proteomics-based analysis can identify protein expression changes and peptide modifications caused by chemical adulterants, providing evidence of specimen manipulation even after the adulterant compound itself has fully degraded.”
The practical implication is clear: a specimen that passes all current validity screens may still carry the molecular fingerprint of tampering. Proteomics brings that fingerprint into view. While the method is not yet universally available at point-of-care or smaller forensic labs, its increasing presence in reference laboratory menus signals where the field is heading.
Expert perspective: What most protocols still miss about adulterant detection
Most program managers believe their adulterant detection is solid because they have strips, SVT, and a lab confirmation process. What they often underestimate is the human layer of the system.
Directly observed collections and regular protocol updates are the two most undervalued controls in any program. You can have the best test chemistry available and still miss a substitution because the collection process had a two-minute window where the donor was unsupervised.
Temperature verification is an example most programs perform correctly in theory and inconsistently in practice. Checking specimen temperature within four minutes of collection is mandatory under federal guidelines, but collection staff under volume pressure often delay or skip this step. That lapse is exactly what sophisticated donors count on.
Protocols must be treated as living documents. Adulteration techniques evolve faster than most standard operating procedures are reviewed. A protocol written two years ago may not address the latest generation of synthetic urine products or the use of enzyme-based adulterants that escape oxidant screens.
The other gap is organizational. Lab professionals and field collection teams often operate in silos. When a lab identifies an unusual adulterant pattern across multiple specimens, that intelligence rarely flows back to the collection site to update observational protocols. Building formal feedback loops between laboratory analysis teams and law enforcement or collection personnel is one of the highest-value process improvements most programs can make. Adulterant detection is a team problem, not a chemistry problem.
Reliable adulterant detection starts with advanced tools
Putting a layered detection protocol into practice requires more than policy updates. It requires tools that are current, validated, and calibrated for the real-world adulteration methods your program faces.
For programs that need broad-panel drug screening paired with built-in adulterant detection, the 22-panel test with adulterants provides simultaneous immunoassay coverage and validity screening in a single collection cup, reducing the risk of procedural gaps between separate tests. If you are building or auditing your current detection framework, our in-depth detection guide walks through product selection, protocol design, and escalation criteria for labs, clinics, and enforcement programs at every scale.
Frequently asked questions
What is the most common way urine specimens are adulterated?
Dilution is most prevalent in drug testing, achieved either by drinking excessive fluids before collection or by adding water directly to the specimen cup at the time of collection.
Are adulterated drug test samples reported as positive?
Yes. Per SAMHSA guidelines, an adulterated sample equals refusal and carries the same consequences as a confirmed positive, including removal from safety-sensitive duties in federally regulated programs.
How can novel adulterants be detected if standard strips miss them?
Advanced methods like mass spectrometry or proteomics detect manipulation signatures in the specimen matrix, including protein and peptide changes caused by chemical adulterants, even if the adulterant compound itself has already degraded.
What does specimen validity testing (SVT) look for?
SVT screens urine for creatinine, pH, specific gravity, and the presence of oxidants or other adulterant markers, establishing whether the specimen is valid and suitable for drug analysis before immunoassay testing begins.