Blog
How quality assurance drives reliable drug testing results

Most laboratory professionals assume their drug testing results are accurate simply because the instruments are running. That assumption is where errors begin. Without a structured quality assurance (QA) program, even well-equipped labs can produce results that fail regulatory scrutiny or, worse, harm patients and compromise legal cases. Proficiency testing catches 92-98% of errors in dual-programmed labs, yet top deficiencies still cluster around PT participation and analytic systems. This guide walks through QA frameworks, core protocols, proficiency testing, and the practical differences between clinical and forensic settings so you can build a program that holds up under any audit.
Table of Contents
- Quality assurance in drug testing: Defining the foundation
- Core QA elements: Method validation, SOPs, and calibration
- Proficiency testing and audits: The QA feedback loop
- Practical QA: Special challenges in clinical versus forensic drug testing
- Best practices for QA implementation and continuous improvement
- Expert-recommended drug test kits and resources
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| QA ensures accuracy | A strong quality assurance framework drastically reduces errors in drug testing laboratories. |
| Regulatory compliance is required | Standards like CLIA and CAP mandate continuous QA through validation, SOPs, and proficiency testing. |
| Clinical and forensic QA differ | Special QA protocols address unique challenges like chain of custody in forensic and interference management in clinical settings. |
| Continuous improvement matters | Regular audits and benchmark-driven PT keep the laboratory’s quality system up to date. |
Quality assurance in drug testing: Defining the foundation
Quality assurance in a drug testing lab is not a checklist you complete once a year. It is a continuous, proactive system designed to prevent errors before they reach a result. QA is preventive and process-focused, while quality control (QC) is reactive and product-based. Both are necessary, but confusing them is a common and costly mistake.
The table below clarifies the distinction:
| Dimension | Quality assurance (QA) | Quality control (QC) |
|---|---|---|
| Focus | Process and system design | Individual test results |
| Timing | Before and during testing | After testing |
| Goal | Prevent defects | Detect defects |
| Examples | SOP development, staff training | Control sample review, calibration checks |
Core QA principles include:
- Standard operating procedures (SOPs): Written, version-controlled instructions for every procedure
- Staff training and competency assessment: Documented and recurring
- Method validation: Confirming assay performance before clinical or forensic use
- Internal and external audits: Scheduled reviews of processes and records
- Corrective and preventive action (CAPA): Formal response to identified gaps
Neglecting QA while relying only on QC is like checking a finished product for cracks without ever inspecting the manufacturing line. CLIA and CAP regulations require labs to maintain documented QA programs, and failure to comply can result in certificate suspension or loss of accreditation. Understanding accuracy in drug test results starts with knowing what CLIA waived means for your testing category and how it shapes your QA obligations.
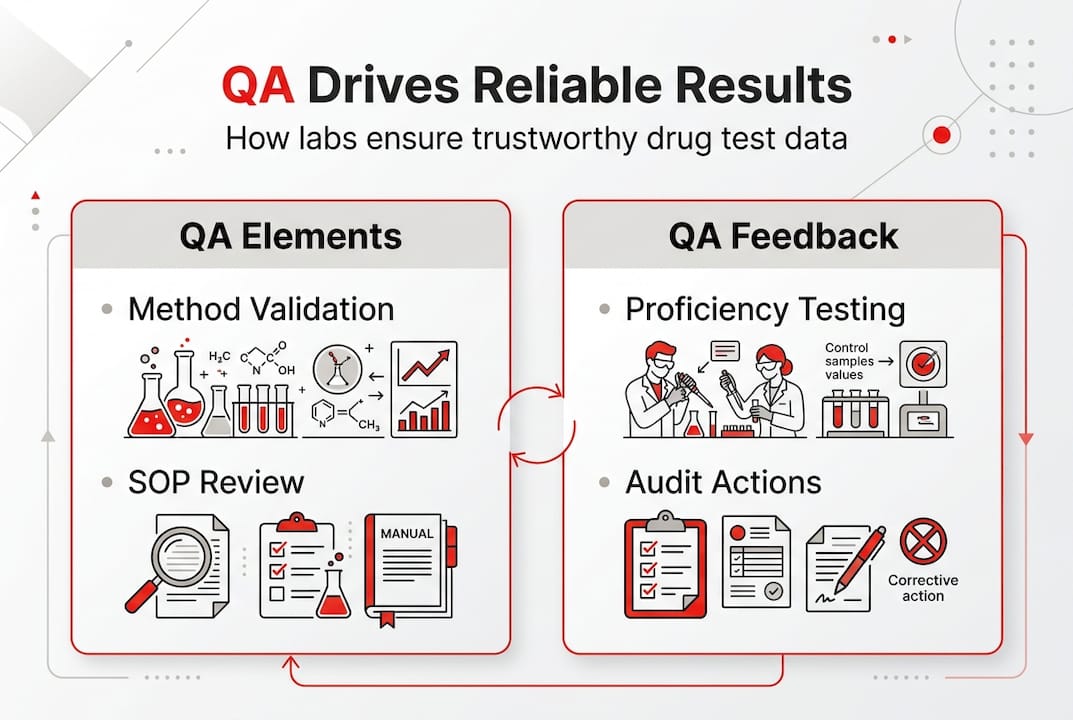
Core QA elements: Method validation, SOPs, and calibration
Implementing QA effectively means building each element deliberately. Skipping steps here creates gaps that auditors and PT programs will find. Key methodologies include validation, SOPs, and equipment calibration as the structural backbone of any compliant lab program.
Here is a practical sequence for implementing core QA elements:
- Define the scope of testing and identify all analytes, matrices, and cutoff concentrations your lab will report.
- Develop and approve SOPs for specimen collection, handling, analysis, and result reporting before any testing begins.
- Validate each method using ICH Q2 guidelines for clinical labs or ANSI/ASB standards for forensic labs, establishing accuracy, precision, limit of detection (LOD), and limit of quantitation (LOQ).
- Calibrate all instruments on a defined schedule, using NIST-traceable reference materials where required.
- Train all staff on validated methods and document competency assessments at hire and annually thereafter.
- Implement CAPA procedures so any deviation triggers a formal review and documented resolution.
- Schedule internal audits quarterly and external audits at least annually to verify ongoing compliance.
Clinical labs use validated LC-MS workflows with calibrated instrumentation to confirm immunoassay screen results, reducing false positives and supporting defensible reporting. Method validation is not a one-time event. Any change in reagent lot, instrument, or procedure triggers revalidation.
Pro Tip: Review and update every SOP at least annually, or immediately after a regulatory change, instrument upgrade, or CAPA event. Outdated SOPs are one of the most cited deficiencies during lab inspections.
Proper documentation for drug testing compliance and a well-mapped random drug testing workflow both depend on these validated, calibrated foundations being in place first.
Proficiency testing and audits: The QA feedback loop
Proficiency testing (PT) is the external benchmark that tells you whether your lab’s results match what other qualified labs produce on the same samples. It is not optional. Labs must participate in PT regularly, with forensic labs required every 180 days and CLIA-regulated labs on a biennial cycle at minimum. Dual PT programming detects up to 98% of errors, making it the single most powerful error-catching tool available.
Dual PT identifies up to 98% of laboratory errors when applied consistently across two independent programs.
The table below summarizes PT requirements and common performance gaps:
| Setting | PT frequency | Target pass rate | Common deficiencies |
|---|---|---|---|
| CLIA clinical lab | Every 6 months per specialty | 80% or higher per event | PT participation (4.39%), analytic systems (5%) |
| Forensic lab | Every 180 days | 100% for reportable analytes | Chain of custody documentation, blind PT gaps |
| CAP-accredited lab | Per CAP schedule | Per analyte-specific criteria | SOP currency, calibration records |
CLIA top deficiencies consistently include PT participation failures and analytic system gaps, meaning many labs are not using the tools they already have access to. Audit cycles reinforce PT findings by reviewing records, interviewing staff, and observing live procedures. Together, PT and audits create a feedback loop: PT identifies what is wrong, audits identify why it is wrong, and CAPA closes the gap.
An efficient drug test quality control workflow integrates PT scheduling, audit timelines, and CAPA tracking into a single operational calendar so nothing falls through the cracks.
Practical QA: Special challenges in clinical versus forensic drug testing
All labs share the same QA principles, but the application differs sharply between clinical and forensic environments. Getting this wrong in a forensic context can invalidate evidence in court. Getting it wrong clinically can lead to incorrect treatment decisions.
Forensic labs require stricter chain of custody and blind PT to prevent bias, while clinical labs must actively manage matrix effects using orthogonal confirmation tests. Here is how the challenges break down:
Forensic lab QA challenges:
- Maintaining unbroken chain of custody from collection through final report
- Using blind PT samples to prevent analyst bias
- Meeting CAP Forensic Drug Testing accreditation requirements
- Documenting every sample transfer with timestamps and signatures
- Defending results in legal proceedings with complete audit trails
Clinical lab QA challenges:
- Controlling for matrix effects in urine, blood, and oral fluid specimens
- Running orthogonal confirmation tests (e.g., LC-MS/MS) after immunoassay screens
- Managing high specimen volumes without sacrificing turnaround time
- Integrating QA into electronic health record (EHR) systems
“CAP Forensic Drug Testing accreditation is not just a credential. It is the operational framework that makes chain of custody defensible in any legal or regulatory proceeding.”
Pro Tip: Apply risk-based thinking to prioritize QA resources. Identify the three highest-risk steps in your workflow, such as specimen receipt, result entry, and report release, and concentrate your controls there first before expanding to lower-risk areas.
A QMS should track CAP, CLIA, and NIST-traceable standards in a unified system so clinical and forensic requirements do not create competing documentation burdens. Use the forensic drug testing checklist and review the importance of forensic drug testing to align your program with current standards.
Best practices for QA implementation and continuous improvement
Building a QA program is one thing. Sustaining it under real operational pressure is another. The labs that maintain consistent compliance share a few common practices that separate them from those that scramble before every audit.
Hybrid QA/QC models and automation enable over 90% error detection, and digital quality management systems (QMS) make tracking corrective actions, calibration records, and PT results far more reliable than paper-based systems. Calibration benchmarks require R2 greater than 0.99 in validated workflows, with robust means and standard deviation of the percent accuracy (SDPA) tracked across every run.
Here is a numbered checklist for ongoing QA improvement:
- Assign a dedicated QA officer or designate a trained staff member with formal QA responsibilities.
- Automate calibration reminders and PT enrollment deadlines inside your digital QMS.
- Conduct unannounced internal audits at least twice per year to simulate regulatory inspections.
- Track key performance indicators monthly: error detection rate, PT pass rate, calibration success rate, and CAPA closure time.
- Review all near-miss events, not just confirmed errors, to catch systemic issues early.
- Update your QA program annually to reflect new regulatory guidance, technology changes, and lessons from PT feedback.
Common pitfalls to avoid:
- Treating SOPs as static documents that never need revision
- Skipping PT enrollment because the schedule feels inconvenient
- Relying on a single QC level instead of running high, low, and negative controls
- Failing to close CAPA items within the defined timeframe
- Underestimating the documentation burden for forensic chain of custody
A well-structured drug testing reporting workflow ties all of these elements together, ensuring that every result leaving your lab is traceable, defensible, and compliant.
Expert-recommended drug test kits and resources
Putting a strong QA program in place requires more than good processes. It requires test kits that are built to perform consistently under those processes. Kits that lack validated cutoffs or reliable reagent stability create QA problems no SOP can fully fix.
At rapidtestcup.com, you will find top urine drug test kits reviewed for accuracy, regulatory compliance, and lab-grade reliability. The 18 panel drug test cup includes adulterant detection and covers a broad analyte panel suited for both clinical and forensic screening programs. If you are still evaluating which format fits your workflow, the guide to selecting drug test kits walks through panel options, CLIA waiver status, and volume considerations so your purchasing decision supports your QA goals from day one.
Frequently asked questions
How does proficiency testing improve accuracy in drug testing labs?
Dual PT programming detects up to 98% of errors, giving labs an external benchmark to identify and correct systematic problems before they affect patient or forensic outcomes.
What is the difference between QA and QC in drug testing labs?
QA is process-oriented and preventive, focused on building systems that stop errors from occurring, while QC is product-oriented and reactive, catching defects in individual test results after the fact.
Which standards are most important for quality assurance in clinical labs?
CLIA and CAP both require QA programs structured around proficiency testing, validated methods, and regular audits, making them the two most operationally significant frameworks for clinical drug testing labs.
How do forensic labs ensure chain of custody for drug testing?
Forensic labs require strict chain of custody documentation at every sample handoff, combined with blind proficiency testing to eliminate analyst bias and keep results legally defensible.