Blog
Understanding bulk drug testing importance in 2026

Bulk drug testing programs face persistent accuracy challenges that many professionals underestimate. False positives plague immunoassay screening at rates exceeding 20%, while complex pharmacokinetics create interpretation pitfalls that can derail compliance monitoring. This guide equips healthcare providers, substance abuse professionals, and law enforcement officials with practical knowledge about specimen selection, testing methodologies, result interpretation strategies, and compliance applications. You will learn how to navigate common testing obstacles, leverage expert toxicologist input, and implement scalable solutions that protect program integrity while meeting regulatory requirements in 2026.
Table of Contents
- Specimen Types In Bulk Drug Testing: Advantages And Limitations
- Testing Methods: Trade-Offs Between Immunoassay And Mass Spectrometry
- Interpreting Bulk Drug Test Results: Challenges And Expert Recommendations
- Practical Applications And Compliance Benefits Of Bulk Drug Testing
Key takeaways
| Point | Details |
|---|---|
| Specimen choice impacts accuracy | Urine offers extended detection windows while oral fluid enables noninvasive collection with shorter timeframes |
| Testing method trade-offs matter | Immunoassay delivers speed but risks false results; mass spectrometry provides superior specificity at higher cost |
| Expert interpretation prevents errors | Toxicologist consultation clarifies pharmacokinetic variability and reduces costly misinterpretation |
| Bulk testing enables compliance | Scalable monitoring supports regulatory requirements across healthcare, treatment programs, and law enforcement |
Specimen types in bulk drug testing: advantages and limitations
Selecting the right specimen matrix forms the foundation of accurate bulk drug testing programs. Urine remains the dominant matrix for drug screening, but oral fluid and other emerging specimens offer new opportunities for noninvasive testing. Each matrix presents distinct advantages and limitations that directly impact detection capabilities, collection logistics, and result reliability.
Urine testing dominates professional settings because established protocols and extended detection windows make it ideal for compliance monitoring. Most drugs remain detectable in urine for 1 to 3 days after use, with some substances lingering up to 30 days depending on usage patterns and individual metabolism. The advantages of urine drug tests include comprehensive detection capabilities and decades of validation research supporting interpretation standards.
Oral fluid testing has gained traction as a less invasive alternative that simplifies collection procedures and reduces privacy concerns. Detection windows typically span 24 to 48 hours, making oral fluid optimal for recent use detection rather than historical consumption patterns. This shorter timeframe limits applicability in some compliance scenarios but offers value when monitoring immediate impairment risks.
| Specimen Type | Detection Window | Invasiveness | Collection Ease | Best Use Case |
|---|---|---|---|---|
| Urine | 1-30 days | Moderate | Requires privacy | Compliance monitoring, broad detection |
| Oral Fluid | 24-48 hours | Low | Simple, observed | Recent use, workplace screening |
| Hair | Up to 90 days | Low | Simple | Long-term history, forensic analysis |
| Blood | Hours to days | High | Requires training | Impairment assessment, clinical settings |
Urine specimen advantages:
- Extended detection windows capture both recent and historical drug use
- Established cutoff levels and interpretation guidelines reduce ambiguity
- Large sample volumes enable multiple test confirmations
- Cost-effective collection and processing for bulk programs
Urine specimen limitations:
- Privacy requirements complicate observed collections
- Adulteration risks necessitate specimen validity testing
- Collection logistics challenge some field environments
Oral fluid advantages:
- Noninvasive collection improves donor acceptance
- Direct observation prevents substitution and tampering
- Reflects recent drug use and potential impairment
- Simplified logistics for mobile testing programs
Oral fluid limitations:
- Shorter detection windows miss historical use patterns
- Lower drug concentrations challenge analytical sensitivity
- Limited validation data compared to urine testing
- Higher per-test costs in some testing scenarios
Pro Tip: Align specimen selection with program objectives by choosing urine for comprehensive compliance monitoring and oral fluid when recent use detection or simplified collection logistics take priority.
Understanding how drug testing trends 2025 influence specimen preferences helps you anticipate regulatory shifts and technological advances that may expand matrix options. Emerging research continues validating alternative specimens, but urine and oral fluid remain the workhorses of bulk testing programs in 2026.
Testing methods: trade-offs between immunoassay and mass spectrometry
Choosing between immunoassay screening and mass spectrometry confirmation represents a critical decision that balances speed, accuracy, cost, and technical complexity. Immunoassay-based screens are vulnerable to false-positive and negative results, while mass spectrometry provides superior specificity and breadth but requires greater technical expertise and infrastructure.
Immunoassay technology dominates initial screening because rapid results and straightforward operation enable high-throughput testing without specialized laboratory infrastructure. These antibody-based tests detect drug classes rather than specific compounds, producing results in minutes using simple procedures. The trade-off comes in specificity limitations that generate false signals from structurally similar compounds, over-the-counter medications, and dietary supplements.
Mass spectrometry delivers definitive identification by measuring exact molecular weights and fragmentation patterns unique to each substance. This precision eliminates cross-reactivity issues and enables detection of synthetic analogs and designer drugs that evade immunoassay panels. However, the technology demands significant capital investment, trained personnel, and longer turnaround times that challenge rapid decision-making needs.
Immunoassay advantages:
- Results available in 5 to 10 minutes for point-of-care decisions
- Minimal training requirements enable non-laboratory personnel operation
- Low per-test costs support high-volume screening programs
- Portable formats accommodate field testing environments
- CLIA-waived options simplify regulatory compliance
Immunoassay limitations:
- Cross-reactivity generates false positives requiring confirmation
- Limited panels miss emerging synthetic drugs and analogs
- Cutoff levels may not detect low-concentration exposures
- Antibody availability constrains new drug detection
Mass spectrometry advantages:
- Definitive identification eliminates false-positive concerns
- Simultaneous detection of hundreds of compounds in single analysis
- Quantitative results support pharmacokinetic interpretation
- Detects novel psychoactive substances and designer drugs
- Lower detection limits capture trace exposures
Mass spectrometry limitations:
- Equipment costs exceed $100,000 for entry-level systems
- Specialized training requirements limit operator availability
- Turnaround times span hours to days versus minutes
- Sample preparation complexity reduces throughput capacity
- Maintenance and consumable costs increase operational expenses
Pro Tip: Implement a two-tier testing strategy using immunoassay for initial screening and mass spectrometry confirmation for positive results to optimize accuracy while controlling costs in bulk testing programs.
The rapid screening methods explained approach balances practical constraints with accuracy requirements by leveraging each methodology’s strengths. Understanding false positives in drug tests helps you anticipate when confirmatory testing becomes essential to protect program integrity and individual rights.
Your testing strategy should reflect program priorities, available resources, and consequences of incorrect results. High-stakes decisions involving employment termination, legal proceedings, or treatment discharge demand mass spectrometry confirmation, while routine monitoring may accept immunoassay screening with selective confirmation protocols.
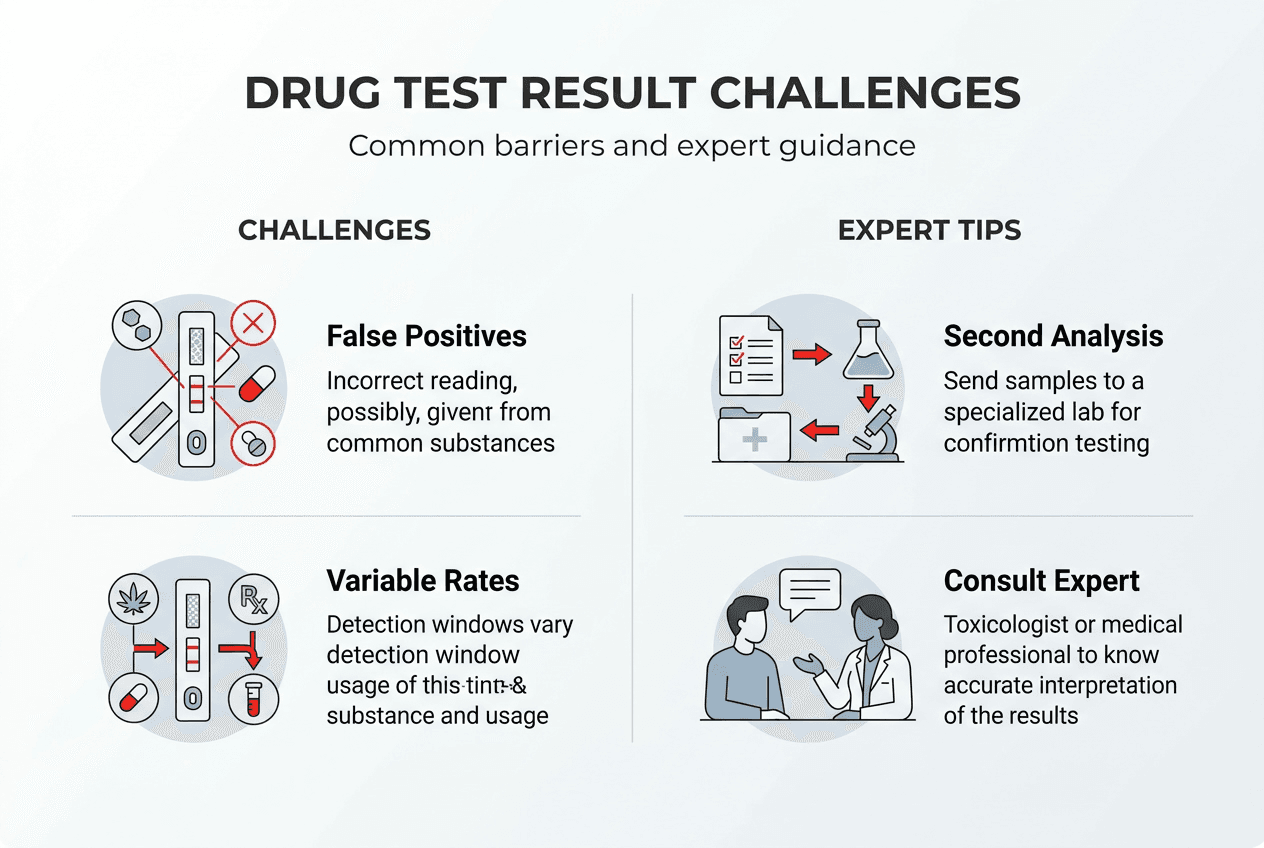
Interpreting bulk drug test results: challenges and expert recommendations
Accurate result interpretation separates effective compliance monitoring from programs plagued by false accusations and missed detections. Result interpretation remains a major challenge due to complex pharmacokinetics and limited clinician training; input from an expert toxicologist is essential to avoid misinterpretation and ensure appropriate follow-up.
Pharmacokinetic variability creates interpretation challenges that confound straightforward positive/negative classifications. Individual differences in metabolism, hydration status, dosing patterns, and drug interactions produce concentration ranges that overlap between compliant therapeutic use and illicit consumption. A positive result may reflect prescribed medication, passive environmental exposure, or deliberate use depending on concentration levels and metabolite patterns.
Misinterpretation risks cascade into serious consequences including wrongful accusations that damage reputations, missed detections that compromise safety, and compliance failures that trigger regulatory sanctions. The complexity demands systematic interpretation protocols supported by toxicology expertise rather than relying on automated result flags or untrained personnel judgment.
Key steps to accurate interpretation and follow-up:
- Review medication history and authorized prescriptions before flagging positive results as violations
- Compare drug concentrations against established cutoff levels and therapeutic ranges
- Analyze metabolite ratios to distinguish direct consumption from passive exposure or medication use
- Consider specimen collection timing relative to last known drug administration
- Evaluate specimen validity indicators including creatinine, pH, and adulterant markers
- Consult toxicologist expertise when results conflict with clinical presentation or program expectations
- Document interpretation rationale to support compliance decisions and potential appeals
Pro Tip: Establish standing consultation agreements with board-certified toxicologists to enable rapid expert review when interpretation questions arise, preventing delays that compromise program operations.
Expert toxicologist review reduces interpretation errors by 75% compared to automated result reporting alone, preventing costly compliance failures and protecting individual rights in high-stakes monitoring programs.
The drug test result interpretation guide provides frameworks for systematic result evaluation, while understanding accuracy in drug test results helps you recognize when additional investigation becomes necessary. Complex cases involving polypharmacy, unusual concentration patterns, or inconsistent results demand expert analysis rather than algorithmic interpretation.
Integrating toxicologist consultation early in your workflow prevents downstream problems by catching interpretation errors before they trigger inappropriate actions. This proactive approach costs less than managing false accusation fallout or regulatory violations stemming from misinterpreted results. Your program’s credibility depends on interpretation accuracy that withstands scrutiny from medical review officers, legal proceedings, and regulatory audits.
Practical applications and compliance benefits of bulk drug testing
Implementing bulk drug testing transforms compliance monitoring from reactive problem management to proactive risk mitigation across healthcare, substance abuse treatment, and law enforcement settings. Scalable testing enables systematic surveillance of large populations while controlling costs and maintaining regulatory compliance in 2026.
Bulk testing programs support federal and state compliance requirements by providing documented evidence of monitoring activities, detection capabilities, and response protocols. Healthcare facilities satisfy Joint Commission standards, substance abuse programs meet SAMHSA grant requirements, and law enforcement agencies fulfill probation supervision mandates through systematic testing protocols.
Key benefits of bulk drug testing implementation:
- Cost savings through volume purchasing reduce per-test expenses by 40% to 60% compared to individual orders
- Rapid turnaround enables timely intervention when results indicate compliance violations or safety risks
- Standardized procedures across large populations improve result comparability and trend analysis
- Inventory management simplifies logistics by consolidating orders and reducing stockout risks
- Quality assurance becomes more consistent when using uniform testing products and protocols
- Regulatory documentation strengthens through systematic record-keeping and audit trails
| Setting | Compliance Requirement | Bulk Testing Role | Key Benefit |
|---|---|---|---|
| Healthcare | Joint Commission monitoring standards | Systematic patient screening protocols | Risk mitigation, safety documentation |
| Substance Abuse Treatment | SAMHSA grant compliance, treatment verification | Regular participant monitoring, outcome tracking | Program effectiveness evidence, funding compliance |
| Law Enforcement | Probation/parole supervision requirements | Offender monitoring, violation detection | Public safety, recidivism reduction |
| Workplace Programs | DOT regulations, safety policies | Employee screening, reasonable suspicion testing | Accident prevention, liability protection |
The bulk drug test ordering process streamlines procurement by consolidating vendor relationships and standardizing product specifications across your organization. This consistency improves training efficiency, reduces user error, and simplifies quality control procedures.
Bulk testing programs generate data that supports continuous improvement through trend analysis and outcome measurement. You can identify emerging drug threats, evaluate intervention effectiveness, and demonstrate program value to stakeholders and regulators. This evidence-based approach strengthens funding justifications and policy development.
Staying current with drug testing trends guide helps you anticipate regulatory changes and technological advances that may require program adjustments. Proactive adaptation maintains compliance while optimizing testing strategies for evolving drug use patterns and detection capabilities.
Get reliable bulk drug testing solutions with RapidTestCup
RapidTestCup delivers comprehensive drug testing products designed specifically for healthcare providers, substance abuse programs, and law enforcement agencies managing large-scale compliance monitoring. Our extensive catalog includes multi-panel test cups, dip cards, and specimen collection supplies that combine accuracy with operational efficiency.
The 12 panel ADLTX cup product features integrated adulterant detection that identifies specimen tampering attempts while screening for common drugs of abuse. This dual-purpose design reduces costs by eliminating separate validity testing while maintaining result integrity. We offer 15-panel and 22-panel configurations to match your specific detection requirements without paying for unnecessary testing capacity.
Our bulk drug test ordering guide simplifies procurement with volume discounts that reduce per-test costs while ensuring consistent product availability. CLIA-waived, FDA-approved products meet regulatory standards without complex laboratory infrastructure requirements. Individual test options like the MET drug test strip enable targeted screening when comprehensive panels exceed program needs.
Frequently asked questions
What is bulk drug testing and why is it important?
Bulk drug testing involves simultaneous processing of multiple specimens using standardized procedures and consolidated procurement to achieve cost efficiency and operational consistency. This approach enables healthcare facilities, treatment programs, and law enforcement agencies to monitor large populations systematically while maintaining compliance with regulatory requirements. Bulk testing reduces per-test costs by 40% to 60% compared to individual orders while improving quality control through standardized protocols. The scalability supports proactive risk management rather than reactive problem response, protecting public safety and program integrity.
How do specimen types affect drug test accuracy in bulk testing?
Specimen selection directly impacts detection timeframes, collection logistics, and result reliability in bulk testing programs. Urine provides extended detection windows spanning 1 to 30 days that capture both recent and historical drug use patterns, making it optimal for comprehensive compliance monitoring. Oral fluid offers noninvasive collection with 24 to 48-hour detection windows suited for recent use assessment and simplified field testing. Each matrix presents trade-offs between invasiveness, detection capability, and operational complexity that must align with program objectives and regulatory requirements.
What are common challenges with immunoassay-based drug screens?
Immunoassay screening methods produce false-positive results when structurally similar compounds, over-the-counter medications, or dietary supplements trigger antibody reactions designed to detect target drugs. Cross-reactivity rates exceed 20% for some drug classes, requiring confirmatory testing to prevent wrongful accusations. False negatives occur when drug concentrations fall below detection thresholds or when novel synthetic compounds evade antibody recognition. Implementing two-tier testing protocols with mass spectrometry confirmation for positive immunoassay results reduces interpretation errors by 75% while controlling costs through selective confirmation strategies.
Why is expert toxicologist input essential in bulk drug testing?
Toxicologist expertise clarifies complex pharmacokinetic patterns that confound automated result interpretation, including individual metabolism variations, drug interaction effects, and concentration ranges that overlap between therapeutic use and illicit consumption. Expert review distinguishes prescribed medication from policy violations, identifies passive environmental exposures, and detects specimen tampering attempts that compromise result validity. Consultation prevents misinterpretation errors that trigger inappropriate compliance actions, regulatory violations, and legal challenges. Standing toxicologist agreements enable rapid expert review when results conflict with clinical expectations or program standards, maintaining operational efficiency while protecting interpretation accuracy.