Blog
Cup vs Dip Card Drug Tests: Key Differences Explained

TL;DR:
- Choosing the correct drug test format impacts accuracy, compliance, and workflow efficiency.
- Cup tests minimize specimen handling and contamination risk, ideal for regulated environments.
- Dip cards offer flexibility and cost savings but require trained staff for accurate results.
Choosing the wrong drug test format is not just an operational inconvenience. It can mean failed audits, compromised chain of custody, or results that don’t hold up under scrutiny. Many professionals in substance abuse programs, law enforcement, and workplace safety assume that rapid immunoassay tests are interchangeable, but the format you choose directly shapes your workflow, your accuracy risk, and your compliance posture. This article breaks down the real differences between cup and dip card drug tests, so you can make an informed, defensible decision for your setting.
Table of Contents
- What are cup and dip card drug tests?
- How do cup and dip card tests compare?
- Selecting the right drug test for your setting
- Common pitfalls and best practices
- What most professionals miss about drug test selection
- Explore advanced drug test solutions
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Cup tests minimize contamination | Integrated collection and result display reduce errors and accidental exposure. |
| Dip cards offer flexibility | Dip cards suit mass screening and budget-conscious environments with easy scalability. |
| Setting determines best method | Workplace, clinical, and law enforcement needs can dictate the most effective testing solution. |
| Avoid common pitfalls | Proper handling and interpretation are essential for compliance and accurate results. |
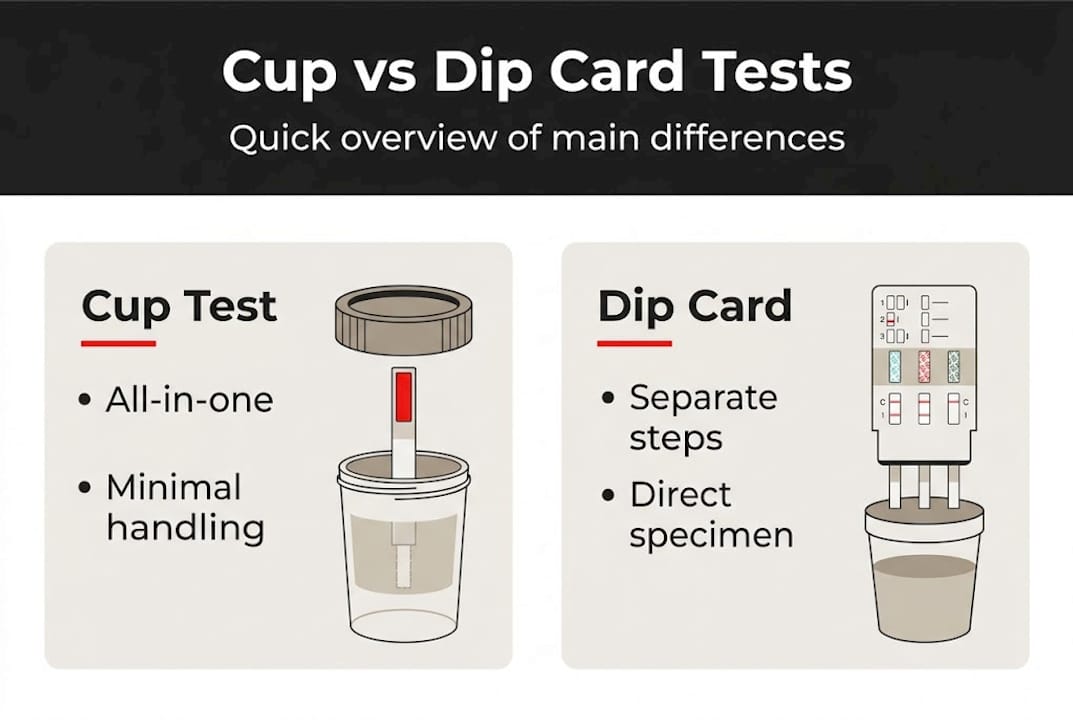
What are cup and dip card drug tests?
Before comparing them, you need to understand exactly what each product does and where it fits in a professional testing workflow.
A drug test cup is an all-in-one specimen collection and testing device. The donor urinates directly into the cup, and the test strips built into the side of the cup react with the sample automatically. No pouring, no transferring, no separate handling of the specimen. Cup drug tests integrate sample collection and automated result display, which significantly reduces the number of steps between collection and result.
A dip card drug test is a separate testing panel that you physically dip into a collected urine specimen. The donor provides a sample in a collection cup, and the technician then submerges the dip card into the sample to produce results. You can explore the role of dip card tests in screening programs more closely to understand where they add value.
Here’s a quick look at common usage scenarios for each:
- Cup tests are widely used in clinical settings, substance abuse programs, and pre-employment screenings where a controlled, streamlined process is required
- Dip cards are frequently used in high-volume or flexible environments where technicians need to test from a single sample across multiple panels, or where cost per test is the primary concern
- Cup tests reduce technician contact with the specimen, lowering contamination and biohazard risk
- Dip cards allow more customization, since you can select specific panels without being locked into a fixed cup configuration
- Both formats can screen for a wide range of substances and both offer CLIA waived, FDA-cleared options
Understanding the cup vs dip card difference comes down to how much direct handling your process requires and how important standardization is to your facility’s documentation.
Pro Tip: If your program operates in a setting where specimen handling must be minimized for liability reasons, cups are the safer structural choice. If you’re running mass screenings where cost per unit matters most and your technicians are highly trained, dip cards can be equally effective.
How do cup and dip card tests compare?
With definitions established, let’s see how cup and dip card tests stack up head-to-head.
| Feature | Cup test | Dip card test |
|---|---|---|
| Specimen handling | Minimal, donor self-collects | Requires technician to dip card |
| Contamination risk | Lower | Moderate |
| Cost per test | Slightly higher | Generally lower |
| Result speed | 5 minutes | 5 minutes |
| Panel customization | Fixed per cup design | Flexible |
| Workflow efficiency | High in controlled settings | High in bulk operations |
| Chain of custody | Easier to document | Requires extra documentation |
Cup tests reduce contamination risk compared to dip cards because the sample never needs to be poured or transferred. That matters in regulated environments where specimen integrity is challenged in court or reviewed by accreditation bodies.
Accuracy and reliability are rated highly for both methods when procedures are followed correctly. The gap between the two formats is rarely about the chemistry. It’s about the process surrounding the test.
Here’s what professionals frequently overlook when evaluating these options:
- Cup tests come pre-assembled, which removes the chance of attaching wrong panels to a sample
- Dip cards can be retested or used individually if one panel is inconclusive
- Dip card reliability depends heavily on proper temperature and time of immersion
- Cup test accuracy benefits from built-in temperature strips that detect sample validity automatically
For busy facilities, the operational burden matters as much as the science. A cup test requires fewer technician decisions per sample, which reduces human error in high-throughput settings. A dip card gives you more control and flexibility, but that flexibility only helps when your staff is trained to use it correctly.
“Cup tests are favored for integrated process and reduced contamination risk; dip cards offer flexibility and cost efficiency.” The right choice is the one that matches both your technical environment and your compliance obligations.
Selecting the right drug test for your setting
After comparing core features, it’s time to apply these differences to real-world settings.
Every professional environment has its own pressures. The right test format is the one that fits your workflow, not just the one that looks good on paper. Use the following factors to guide your selection:
- Volume of tests per day. High-volume settings benefit from the efficiency of cup tests. If you’re processing dozens of specimens daily with limited staff, reducing handling steps protects both your staff and your results.
- Level of technician training. Dip cards require more hands-on steps. If your team is experienced and well-trained, dip cards can be cost-effective. For newer or rotating staff, cups reduce the risk of procedural errors.
- Compliance documentation requirements. If your program is subject to regulatory review, courts, or accreditation, the built-in chain of custody features of a cup format simplify your documentation process significantly.
- Budget constraints. Dip cards typically cost less per unit. If budget is tight and you have the staff training to compensate, they’re a legitimate option for certain programs.
- Panel requirements. If you need to test for a broad or unusual combination of substances, drug test cup advantages include multi-panel configurations that streamline testing for common substance combinations.
Healthcare settings often prefer test cups for faster processing and reduced handling, especially in substance abuse treatment programs where patient throughput and result accuracy both matter.
For law enforcement, custody situations often demand airtight documentation and minimal specimen exposure. Cups are almost always the better fit there. For workplace safety programs in low-risk industries, dip test accuracy combined with lower cost can make dip cards an attractive option for ongoing monitoring.
Pro Tip: Before finalizing your format choice, map out your current testing workflow on paper. Identify every step where a technician touches the sample. Each of those steps is a potential error or documentation gap. The format that eliminates the most of those steps is usually the right one for your setting.
Common pitfalls and best practices
To help you avoid costly mistakes, here’s what you need to know about pitfalls and getting reliable results.
Even well-run programs make avoidable mistakes with rapid drug tests. The consequences range from minor inconveniences to serious compliance failures. Incorrect handling or misinterpretation of rapid tests can lead to compliance failures that undermine your entire program.
Here are the most common errors across both formats:
- Ignoring temperature strips. Both formats include temperature indicators, and skipping this check opens the door to specimen substitution
- Reading results too early or too late. Both cup and dip card tests have defined reading windows, usually 5 minutes. Reading outside that window produces invalid results
- Improper storage. Tests stored in environments outside their rated temperature range degrade before use, affecting sensitivity
- Misidentifying faint lines. A faint line is a negative result in immunoassay testing. This is one of the most misunderstood aspects across all formats
- Skipping documentation. Verbal reports are not sufficient. Photographing or logging results at the time of reading is essential for any compliance-driven program
For best practices around urine collection procedures, establishing a written standard operating procedure that your staff follows without exception protects both your program and your results.
Key compliance checkpoints to build into your process:
- Verify lot numbers and expiration dates before every test batch
- Train staff specifically on result interpretation, not just collection
- Store all test kits according to manufacturer specifications
- Document chain of custody from collection through result recording
Studies on point-of-care testing indicate that procedural errors, not test sensitivity, are responsible for the majority of discordant results in rapid drug screening programs. Accuracy starts before the test strip ever contacts the specimen.
What most professionals miss about drug test selection
Here’s an uncomfortable truth: most facilities pick their drug test format based on habit, not evidence. Someone in a previous role used cups, so the program uses cups. Or a vendor pushed dip cards because of price, and the decision stuck. Neither of those is a compliance strategy.
The smarter framing is to treat test selection as a risk management decision. Ask which format reduces your exposure to documentation failures, misinterpretation, and legal challenge. That reframing changes the conversation entirely.
We’ve seen programs switch from cups to dip cards to cut costs, only to absorb the difference in retraining costs and audit corrections. Explore dip card insights and weigh them against drug cup perspectives with your specific risk profile in mind. The cheapest test per unit is rarely the cheapest test when you factor in error rates and compliance overhead.
Balancing cost and reliability is possible, but only when the format decision is grounded in your actual workflow rather than vendor convenience.
Explore advanced drug test solutions
If your program is ready to tighten its testing process, the right products make a measurable difference. Whether you’re running a substance abuse program, managing workplace safety compliance, or supporting law enforcement screening, having the right format in hand matters.
For cup-based programs, the ADLTX cup offers a reliable 12-panel configuration with built-in adulterant detection. If your workflow calls for dip cards, the 12-panel dip card delivers cost-effective flexibility without sacrificing accuracy. For programs that need broader coverage, the 22-panel test covers an expanded substance range including adulterants and emerging compounds. All products are CLIA waived and FDA approved, shipped fast and ready for professional use.
Frequently asked questions
Which is more accurate, cup or dip card drug tests?
Both deliver reliable results when used correctly, but cups reduce contamination risk thanks to their integrated design, which limits technician contact with the specimen.
Can dip cards be used for workplace drug testing?
Yes. Dip cards are cost-effective and well-suited for mass screening in workplace environments where trained staff handle collection and interpretation.
What factors should I consider when choosing between a cup and dip card?
Choose test type based on your contamination risk tolerance, required speed, compliance documentation needs, and the training level of your staff.
Are there any compliance issues unique to cup or dip card tests?
Compliance depends on proper handling, accurate interpretation, and consistent documentation. Both formats must meet the same regulatory standards, but cups make documentation simpler by design.