Blog
What is a lab-based test? Reliable methods explained

TL;DR:
- Lab-based drug testing involves a structured, process-driven approach that relies on certified equipment and thorough documentation to produce legally and clinically defensible results. Proper management of preanalytical, analytical, and postanalytical phases is essential to prevent errors, with confirmatory testing necessary for accurate, court-proof outcomes. Adhering to regulatory standards and maintaining operational discipline are critical to ensuring reliable and compliant testing programs.
Most professionals assume that running a drug test is straightforward: collect a sample, get a result, and move on. That assumption is exactly where errors begin. Lab-based testing is not a single step but a structured process built on certified equipment, documented protocols, and method-specific standards that directly determine whether your results will hold up under clinical, legal, or regulatory scrutiny. This guide walks through how lab-based testing works, which methods apply to different settings, and what every lab, clinic, or substance abuse treatment center needs to understand to make results count.
Table of Contents
- What is a lab-based test?
- The critical phases of lab-based testing
- Lab-based drug testing methods: Screening vs. confirmatory
- Regulatory requirements: Panels, protocols, and certified labs
- Our perspective: What most guides miss about lab-based testing
- Next steps: Reliable panels and lab testing supplies
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Labs use standard protocols | Lab-based tests follow regulated workflows for accuracy and reliability. |
| Method selection matters | Choosing between screening and confirmatory methods impacts speed, cost, and response to test results. |
| Regulations guide testing | Workplace and clinical labs must use certified panels and proper reporting standards. |
| Error control is essential | Managing errors in every test phase is vital for defensible, reliable drug test results. |
What is a lab-based test?
A lab-based test is not simply any test that happens in a lab. According to MedlinePlus, a lab-based test analyzes a sample in a designated lab to determine clinical values or analyte presence. That distinction matters because it separates controlled, certified analysis from point-of-care rapid testing, which produces results immediately at the collection site but with fewer verification steps.
Common specimen types in lab-based drug testing include:
- Urine: The most frequently used specimen, especially in workplace and substance abuse programs, because it provides a detection window of several days for most substances
- Blood: Used when precise, real-time drug concentration is needed, such as in clinical toxicology or medical emergencies
- Oral fluid: Growing in use for recent exposure detection in roadside or workplace settings
- Hair: Used for long-term exposure history, with a detection window extending up to 90 days
- Tissue: Employed in forensic cases, particularly post-mortem toxicology
“Lab-based testing is process-driven and thoroughly documented, unlike rapid testing, which sacrifices rigor for speed.”
The foundational difference between lab-based and rapid testing is not just location. It is process depth. Rapid tests offer quick results but limited confirmation capability. Lab-based testing follows structured clinical testing workflows that include validation controls, reagent verification, and multiple review points. This is why results from certified labs carry weight in court, during treatment evaluations, and in federal compliance reporting.
Maintaining a disciplined drug test quality control workflow is what separates a lab that can defend its results from one that cannot. That process discipline starts before the sample even reaches the analyzer.
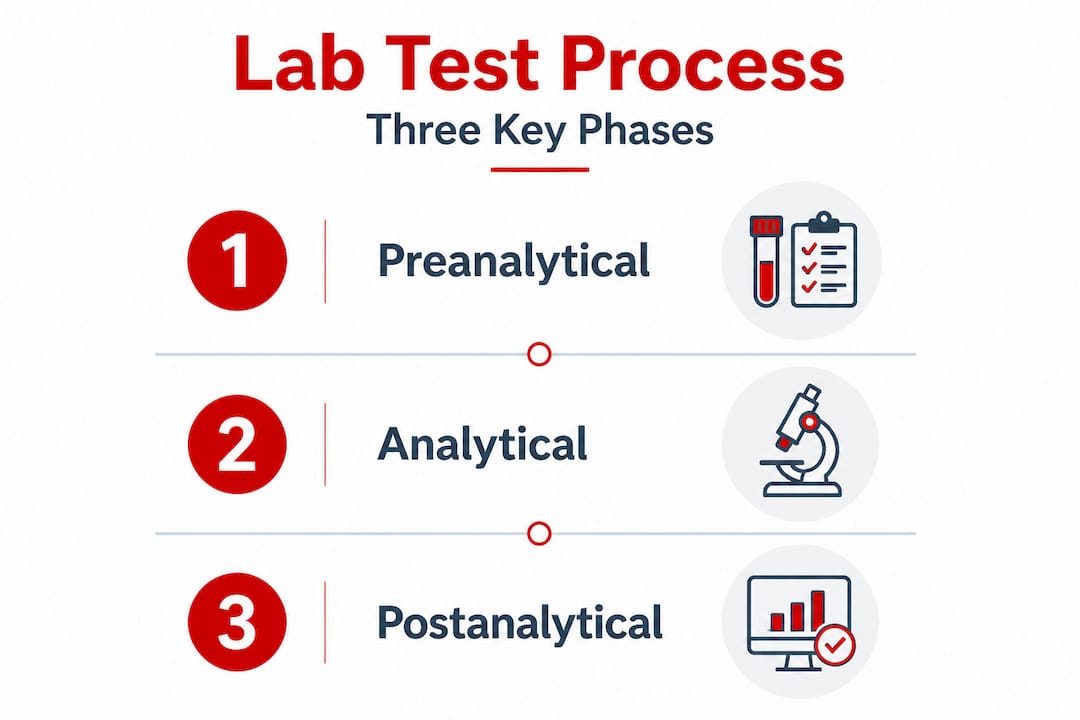
The critical phases of lab-based testing
Reliable lab testing depends on quality control during preanalytical, analytical, and postanalytical phases. Understanding each phase tells you exactly where errors are most likely to occur and what you can do to prevent them.
1. Preanalytical phase
This phase covers everything before the sample reaches the instrument: specimen collection, labeling, storage temperature, chain-of-custody documentation, and transport. It sounds routine, but preanalytical errors account for the majority of lab mistakes. A mislabeled sample, improper storage temperature, or a broken chain-of-custody form can render an otherwise perfect test result unusable.
2. Analytical phase
This is where the actual measurement occurs. It includes selecting the right test method, calibrating instruments, running quality control samples, and executing the assay. Equipment must be properly maintained and calibrated before every run. Controls at known concentrations must pass before patient samples are processed. Any deviation must be documented and investigated.
3. Postanalytical phase
Once results are generated, they must be interpreted correctly, compared to reference ranges or cutoff thresholds, and reported in the proper format. For regulated drug testing, this includes using specific terminology and routing results through a Medical Review Officer (MRO) when required. Errors here, such as miscommunicating a positive result or failing to flag an inconclusive finding, can have serious downstream consequences for patients, employees, and programs.
Pro Tip: Audit each phase independently on a quarterly basis. Most labs treat quality assurance as a single review, but phase-specific audits catch the blind spots that cross-functional reviews miss.
“Errors in the preanalytical phase are the most common and frequently the least monitored — which makes them the most dangerous.”
Reviewing your drug testing reporting workflow against these three phases is a practical way to locate process gaps before they produce a challenged result. Equally important is building a clinical drug test workflow that assigns clear ownership to each phase, because shared responsibility often means accountability falls through the cracks. Industry resources on laboratory workflow examples can also provide benchmarks for how high-performing labs structure these responsibilities.
Lab-based drug testing methods: Screening vs. confirmatory
Not all lab-based drug tests work the same way. Drug testing relies on screening methods such as immunoassays and more definitive confirmatory methods like GC-MS or LC-MS/MS, each with different specificity, cost, and turnaround time. Choosing the wrong method for your context is a serious operational mistake.
| Feature | Screening (immunoassay) | Confirmatory (GC-MS/LC-MS/MS) |
|---|---|---|
| Speed | Fast (minutes to hours) | Slower (hours to days) |
| Cost | Low | Higher |
| Specificity | Moderate | Very high |
| False positive risk | Present | Minimal |
| Legal defensibility | Limited | Strong |
| Common use | Initial screening, large volume | Confirmation, legal, clinical action |
Screening methods use immunoassay technology, where antibodies react with drug metabolites. These tests are efficient for processing large volumes at low cost. However, cross-reactivity is a real concern. Certain over-the-counter medications, herbal supplements, and foods have been documented to trigger false positives. A positive immunoassay result alone is not sufficient to take clinical, legal, or employment action.
Confirmatory methods such as gas chromatography-mass spectrometry (GC-MS) and liquid chromatography-tandem mass spectrometry (LC-MS/MS) identify substances by their molecular structure and mass. This approach leaves virtually no room for ambiguity. Results identify the exact compound and its concentration, which is why confirmatory testing is required in federally regulated workplace programs and in any situation where results drive significant decisions.
Key situations where confirmatory testing is non-negotiable:
- Legal proceedings or forensic investigations
- Employment termination or denial of federal positions
- Patient treatment decisions involving opioid management or addiction medication
- Any positive result disputed by the donor
- Child custody or court-ordered substance monitoring
Understanding the role of confirmatory drug testing in your program design is not just a technical decision. It is an ethical and legal one. Refer to established guidelines on confirmatory testing in substance abuse contexts for detailed protocol frameworks.
Pro Tip: Always document the rationale for your method selection in program policy. When results are challenged, the ability to show that your method choice was intentional and standards-based is a strong line of defense.
Regulatory requirements: Panels, protocols, and certified labs
Lab-based drug testing does not operate in a vacuum. Federally regulated U.S. workplace drug testing programs require authorized panels and formal reporting nomenclature in HHS-certified labs as defined by SAMHSA. These requirements exist to ensure that results produced in one lab carry the same meaning and defensibility as results produced in another.
Key regulatory standards that affect your program:
- SAMHSA guidelines: Define which analytes must be tested, cutoff concentrations for initial screening and confirmation, and reporting standards
- HHS certification: Labs performing federally mandated workplace testing must hold Department of Health and Human Services certification, issued through the National Laboratory Certification Program (NLCP)
- MRO review: A licensed Medical Review Officer must review all positive, adulterated, substituted, or invalid results before they are reported to the employer
- Chain of custody: Federal Form (CCF) documentation must accompany every federally regulated specimen from collection to reporting
| Regulation | Requirement | Who it applies to |
|---|---|---|
| SAMHSA panel | Specific analytes and cutoffs | Federal workplace programs |
| HHS lab certification | Certified facility for analysis | Federal testing labs |
| MRO review | Licensed physician oversight | All federal positive results |
| Chain of custody | Documented specimen handling | All regulated specimens |
Statistic to note: The 2026 Federal Register notice on workplace drug testing updates confirms ongoing revisions to authorized panel lists and reporting language, meaning your program documentation must be reviewed at least annually to remain compliant.
Non-federally regulated programs (such as those run by private employers, clinics, or treatment centers) are not bound to SAMHSA panels but benefit significantly from following them as a baseline. Using panels and cutoffs outside of established norms creates legal exposure and reduces the credibility of your results.
Connecting your standard operating procedures to a workplace drug screening compliance framework is the most efficient path. Resources covering workplace drug testing procedures can help you map current practice to these standards. Cross-referencing an external lab compliance checklist is also a productive step before annual program reviews.
Our perspective: What most guides miss about lab-based testing
Here is what most technical guides do not say plainly: the assay is the easy part. Modern immunoassay platforms and confirmatory instruments are reliable and well-validated. The actual vulnerability in most lab-based drug testing programs is not the chemistry. It is the people, the paperwork, and the process gaps on either side of the instrument.
We see this pattern repeatedly: organizations invest heavily in top-tier analyzers while their specimen labeling procedure is done from memory, their transport log has not been updated in two years, and their reporting template still uses outdated terminology that will not hold up under MRO review. The instrument produces a perfect result, and then a documentation error makes it unusable.
As testing workflow research at NCBI confirms, the mechanical steps of collection, method choice, and interpretation are only as reliable as their weakest link. Error control is what makes a program defensible, not just accurate.
The second blind spot is complacency around postanalytical review. Once a number comes off the machine, teams tend to move on quickly. But interpretation requires context. A result near a cutoff threshold, for example, warrants a different response than one far above it. Reporting a borderline positive without flagging its proximity to the cutoff is technically compliant but operationally weak.
Our recommendation is direct: treat your drug testing program as a system, not a series of individual tasks. That means regular written audits of all three workflow phases, clear role assignments, and scheduled reviews of your regulatory standing. The path to streamline your drug testing process runs through operational discipline, not just equipment upgrades.
Next steps: Reliable panels and lab testing supplies
If this guide has made one thing clear, it is that reliable lab-based drug testing requires the right supplies matched to your specific workflow and regulatory requirements. Generic or uncertified collection tools introduce risk at the one phase you can control completely: collection.
At rapidtestcup.com, we stock CLIA waived, FDA-approved multi-panel drug test cups, urine dip cards, and specimen collection supplies built for professional environments. Whether you run a high-volume substance abuse treatment program, a clinical toxicology workflow, or a federally regulated workplace testing operation, you will find panels and products that match your testing context. We also offer bulk pricing, custom quote options, and fast shipping so your program never stalls waiting on supplies. Browse our complete catalog and find the panels and collection tools your workflow requires.
Frequently asked questions
What’s the difference between lab-based and rapid drug tests?
Lab-based tests are conducted in certified labs with strict protocol controls, offering higher specificity and the option for confirmatory methods than rapid, point-of-care tests, which prioritize speed over analytical rigor.
Why is confirmatory drug testing important?
Confirmatory testing provides definitive, compound-specific results that are legally and clinically defensible. Methods like GC-MS and LC-MS/MS identify exact substances and concentrations rather than just reactive patterns.
What samples are commonly used in lab-based drug testing?
Urine, blood, or tissue samples are commonly used depending on the analytes of interest and the clinical or forensic context of the test.
How do regulations affect lab-based drug testing?
Federally regulated drug testing requires labs to use authorized panels, defined cutoff levels, and standardized reporting formats, all of which protect the legal and clinical defensibility of every result your program produces.